What is neutralization?
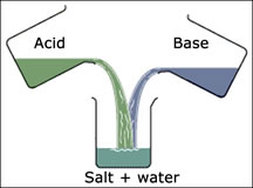
- Neutralization is the process by which an acid and base combine to form water and a salt
What are the four reactions that neutralize an acid?
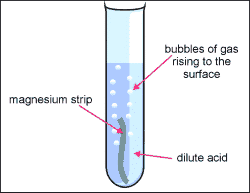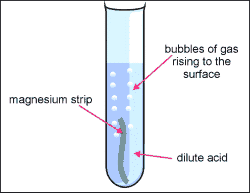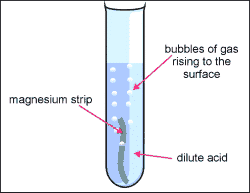
1. Metal + acid = salt + hydrogen gas
- Acid + metal = NaCl + H2
- Acid + carbonates = NaCl + H2O + CO2
- Acid + Base = NaCl + H20
- Acid + Alkaline = NaCl + H20
As you can tell from above salts are products of neutralization.
Here is a list of the salt families produced:
- Hydroxide
- Chlorides
- Nitrides
- Phosphates
- Oxides
Here is an example of a neutralization reaction that produces salts:
- Mg + H2SO4--> H2 + MgSO4
- Magnesium + sulphuric acid = magnesium sulphate
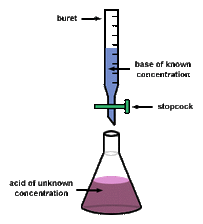
Titrations
- The most common used way to complete a neutralization reaction is through titration
- Titrations is a process in which a solution is added to another solution until the solution is neutralized
- The reason titration's are pink is because it is an indicator and when it neutralizes it turns white