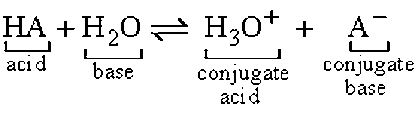Conjugate Acids and Bases
What are conjugate acids?
- When an acid reacts it becomes a conjugate base
- In other words a conjugate acid is a substance created after the base has received a H+ proton
What are conjugate bases?
- When a base reacts it becomes a conjugate acid
- In other words a conjugate base is a substance left behind after the acid has lost a H+ proton
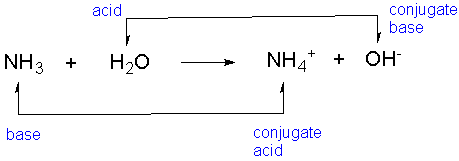
Here's two examples:
So how do you know what to label what?
- It is easiest to first identify which is the acid and which is the base since acids are donors and bases are acceptors as learned in the Bronsted-Lowry Theory
- Acids and bases will always be the reactants so they will be on the left side of the equation
- To identify the acid you look at which compound has the smaller number of hydrogen ions. In the equation above we can tell that H2O has only 2 hydrogen ions where has NH3 has 3 hydrogen ions
- From this we can conclude that the acid is H2O since it is the donor and NH3 since it is the acceptor
- Conjugate acids and conjugate bases are the products always on the right side of the equation
- Lets take a look at this equation: NH3 + H2O --> NH4 + OH-
- H2O which is the acid, has 2 hydrogen ions
- Since OH- has 1 hydrogen ion they differ by a single H and a single charge because OH- is negative
- Since it has a negative charge this means that it lost a hydrogen ion in order to get there
- Following the Bronsted-Lowry theory since H20 is ready to donate a proton. This is considered the acid and since OH- is ready to accept a proton this is the conjugate base
- It is very similar for bases and conjugate acids, since NH3 and NH4+ differ by a single proton and single charge. Since NH3 has 3 hydrogen ions and is missing a proton, NH4+ has it and is ready to donate the proton
- Since NH3 is ready to accept the proton this makes it the base and NH3 the base. Again this follows the Bronsted Lowry Theory because the definition of bases are proton acceptors and NH3 is ready to accept the proton